Secondary hydroxyl groups of the epoxy resin backbone may be reacted with polyisocyanates, aromatic as well as aliphatic, in two — component ambient curing systems that are commonly known as epoxy-urethane hybrid systems. This type of coating combines advantages of epoxy and polyurethane systems.
2.9.3.3 Carboxylic acids and anhydrides
|
Figure 2.54: Curing of epoxy with (a) carboxylic acid and (b) anhydride |
In the presence of amine catalysts, carboxylic acids react with epoxy groups, producing hydroxyl esters at elevated temperatures (Figure 2.54). This type of curing chemistry is used in epoxy-polyester hybrid powder coatings. Some examples of such polymers are carboxylic acid terminated polyesters or carboxylic acid functional acrylic resins. Self-cross-linking acrylic resins containing both (meth)acrylic acid and glycidyl methacrylate in the same copolymers, are also known.
Cyclic anhydrides of carboxylic acid are also used as cross-linkers for epoxy resins. Curing is initiated by reaction of a hydroxyl with an anhydride to yield a half ester and a carboxylic acid group. The newly formed carboxylic acid will in turn react with the epoxy to generate an ester and a new hydroxyl group. These complementary reactions result in a cross-linked network. Etherification reactions
(side reactions) between epoxy-epoxy and epoxy-hydroxyl groups are also known to contribute to a cross-linked network. Examples of some typical cyclic anhydrides are pyromellitic anhydride, hexahydrophthalic anhydride, methyltetrahydrophthalic anhydride and ethylene glycol bis(anhydromellitate). These systems are more sensitive to alkali than amine cured epoxy systems.
 12 октября, 2015
12 октября, 2015  Pokraskin
Pokraskin 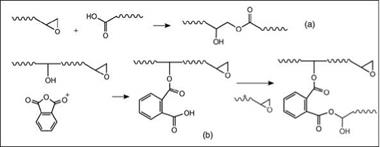
 Опубликовано в рубрике
Опубликовано в рубрике 