This is the most commonly used route in alkyd resin manufacturing. Oils, being cheaper raw materials as compared to corresponding fatty acids, are naturally preferred for alkyd resins, but the oil does not react readily with acid or alcohol. Therefore, to avoid a heteroge-
|
|
neous mixture of unmodified polyester and unreacted oil, first the oil is reacted with a polyol by transesterification (alcoholysis) in the presence of a basic catalyst (such as sodium hydroxide or lithium hydroxide) into a hydroxyl functional intermediate, monoglyceride. The monoglyceride is then reacted with phthalic anhydride and other polybasic acids to form alkyd resins. Generally, the alcoholysis product of oil and polyol contains a mixture predominantly containing a-monoglyceride and b-monoglyceride with some diglyceride, triglyceride and glycerol. The simplest possible representation of the process is shown in Figure 2.8.
 29 августа, 2015
29 августа, 2015  Pokraskin
Pokraskin 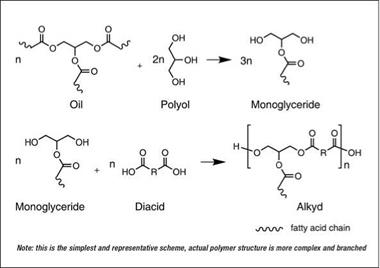
 Опубликовано в рубрике
Опубликовано в рубрике 