The electrical conductivity of carbon blacks is inferior to that of graphite, and is dependent on the type ofproduction process, as well as on the specific surface area and structure. Since the limiting factor in electrical conductivity is generally the transition resistance between neighbouring particles, compression or concentration of pure or dispersed carbon black plays an important role. Special grades of carbon black are used to equip polymers with antistatic or electrically conductive properties. Carbon blacks with a high conductivity and high adsorption capacity for electrolyte solutions are used in dry-cell batteries.
4.1 Chemical Properties 167
4.1.6
Light Absorption
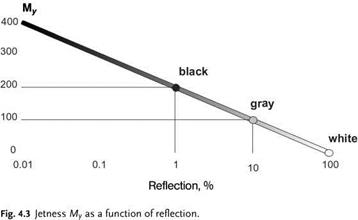 |
The wide use of carbon blacks as black pigments is due to their absorption of visible light. The absorption rate can reach 99.8%. The black color can take on a bluish or brownish tone, depending on light scattering, wavelength, the type of carbon black, and the system into which the carbon black is incorporated. Infrared and ultraviolet light is also absorbed. Therefore, some carbon blacks are used as UV stabilizers in plastics (Figure 4.3).
4.2
Chemical Properties
The general chemical composition of carbon black according to elemental analysis is within the following limits:
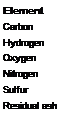 Content (wt.%)
Content (wt.%)
95-99.5
0.2-1.3
0.2-0.5
0-0.7
0.1-1.0
<1
The composition depends on the manufacturing process, the raw material, and the chemical aftertreatment. The ash content of most furnace blacks is <1 wt.% .The ash components can result from the raw material, the salts that are injected to control the structure, and the salts of the process water. The ash content of gas blacks is less than 0.02%.
The surface of carbon blacks contains certain amounts of polynuclear aromatic substances. These are strongly adsorbed and can only be isolated by continuous extraction with solvents, e. g., boiling toluene. For most industrial carbon blacks, the amount of extractable material is below the limit defined by the food laws.
The oxygen content of carbon blacks is of the greatest importance for their application. Oxygen is bound to the surface in the form of acidic or basic functional groups. The amount of surface oxides and their composition depend on the production process and an eventual aftertreatment. Furnace blacks and thermal blacks, which have been produced in a reducing atmosphere, contain about 0.2-2 wt.% oxygen in the form of almost pure, basic surface oxides. Gas and channel blacks, which are manufactured in the presence of air, contain up to 8 wt.% oxygen. In this case, the greater part of the oxygen is contained in acidic surface oxides and only a small portion in basic oxides. The amount of acidic surface oxides can be increased by oxidative aftertreatment. Oxygen contents of up to 15 wt.% may be obtained.
The surface oxides are destroyed at high temperatures. Due to this fact the weight loss at 950 °C (“volatiles”) is a rough indication of the oxygen content of a carbon black. The pH value measured in aqueous slurry is another indication of the degree of oxidation. In general, the pH value is >7 for furnace blacks (low oxygen content, basic surface oxides), 4-6 for gas blacks, and 2-4 for oxidized carbon blacks (high oxygen content, acidic surface oxides with a high amount of polar functional groups). Further organic reactions, e. g., alkylation, halogenation, esterification, can be carried out with the surface oxides to modify the surface properties.
Concerning their oxidation behavior, industrial carbon blacks do not spontaneously ignite when stored in air at 140 °C according to the IMDG Code [4.5]. When ignited in air, carbon black glows slowly. In contrast to coal, dust explosions are not observed under normal test conditions [4.6]. However, ignition sources of extremely high energy, e. g., a gas explosion, may induce a secondary dust explosion in air.
4.3
 30 ноября, 2015
30 ноября, 2015  Pokraskin
Pokraskin  Опубликовано в рубрике
Опубликовано в рубрике 