The coordination number of the chelated metal atom determines the number of linkages to functional groups. It is typically greater than the valency of the metal ion; for example, the divalent ions of copper and nickel have coordination numbers of four, and the trivalent ions of chromium, cobalt, iron a coordination number of six. In the case of iron the coordination number six applies for the di — and trivalent forms.
The most important chelate types are illustrated in Figure 2.7. Compounds in column A display a metal ion with coordination number four. The coordination number six is represented by the chelates of column B and C. Rows a, b, and c cover bi-, tri — and tetrafunctional or — dentate ligands, respectively. The types Ab and the types of column B contain additional monofunctional ligands to complete the coordination numb ers of four and six, respectively.
As far as dyes are concerned, chelates of type Cb are termed 1:2 or symmetrical 1:2 metal complexes if the two tridentate ligands are equal, and mixed or unsymmetrical 1:2 metal complexes in the other case. Chelates of type Bb represent 1:1 metal complexes. The types Cb and Bb include, in general, azo and azo- methine metal complex dyes, whereas chelates of the quadridentate types Ac and Bc are derived predominantly from formazan and phthalocyanine chromophores.
|
Figure 2.7 Types of chelates. M = central metal atom, L = monofunctional ligand, X-X = bidentate ligand, X-X-X = tridentate ligand, X-X-X-X = tetradentate ligand. |
Stability. Ligands with two or more donor groups afford monocyclic or polycyclic — cyclic metal chelates that result in enhancement of the complex stability relative to two or more mono functional nucleophiles. The increasing tendency to form complexes with increasing number of donor groups per ligand, known as the chelate effect, was discovered by Schwarzenbach [22] and is discussed in [23].
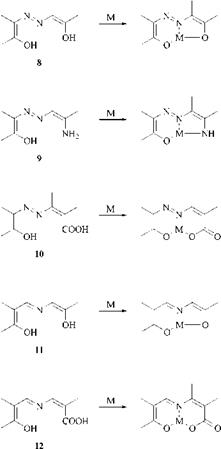
Thus, o, o’-dihydroxyazo (8), o, o’-hydroxyaminoazo (9), o, o’-carboxyhydroxy — azo (10), o, o’-dihydroxyazomethine (11), and o, o’-carboxyhydroxyazomethine (12) residues yield with metals stable annelated 5,6- and 6,6-membered ring systems, respectively (Scheme 2.13). The residues in the azo compounds are in most cases benzene, naphthalene, pyrazolone or open-chain enol-keto systems, while in the azomethine compounds they are mostly benzene systems.
|
Scheme 2.13 Tridentate ligands and their metal chelates (M = Cu2+, Cr 3+, Co3+, etc) |
Chelates in which one nitrogen atom of the azo group and that of the azomethine group form part of the coordination system can also be called medially metallized azo and azomethine dyes [24]. The bond joining metal atoms to the nitrogen, sometimes referred to as a secondary valence, may be represented by an arrow or dotted line. The latter is preferred here.
The stability of a metal complex is related to the formation of strain-free five- or six-membered rings, to the number of such chelate rings per ligand, to the basicity of the donor groups and the nature of the metal ion. Chromium(iii) ions impart the greatest stability in the field of azo dye complexes. An in depth description of complex stability is given in [25].
Chelation of a chromphore can also result in a change in properties, including absorption spectra, brilliance, charge, solubility, and affinity for substrates. In general, metallization leads to a bathochromic shift and leads to a distinct improvement in lightfastness and in many cases an enhanced wetfastness compared to the metal-free compound.
Stereochemistry and Isomerism. Metals having a coordination number of four usually give square-planar complexes, whereas metals having a coordination number of six are octahedrally surrounded by the functional groups of ligands.
From the viewpoint of stereochemistry the most interesting metal complexes are the octahedrally coordinated 1:2 chromium and cobalt complex dyes, which are medially metallized azo and azomethine compounds with functional groups in the o — and o’-positions. Three types of isomerism can be discriminated: geometrical, N — аД and that arising from azo-hydrazone tautomerism.
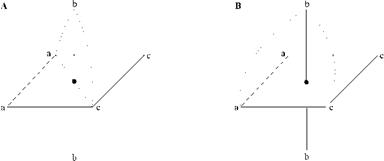
Geometrical isomerism of 1:2 chromium and cobalt complexes is due to the arrangement of the two tridentate dye ligands in a meridial (mer) and a facial (fac) mode (Figure 2.8). The mer configuration is is also known as Drew-Pfitzner structure, after Drew [26] and Pfitzner [27] who independently proposed that the two tridendate ligands are arranged mutually perpendicular. This configuration permits only one pair of enantiomers, which were resolved by Pfeiffer and Saure into optically active forms [28]. The perpendicular arrangement has been verified by X-ray diffraction studies [29].
In the fac configuration, also called the Pfeiffer-Schetty structure, nine isomers are possible: four pairs of enantiomers and a centrosymmetric structure. Since the ligands are arranged parallel to one another, they sometimes are called sandwich complexes. The two mer and the nine fac orientations are depicted in [30].
|
Figure 2.8 Geometrical isomers. A) Meridial arrangement (Drew-Pfitzner structure). B) Facial arrangement (Pfeiffer-Schetty structure). |
The fac isomers were isolated by Schetty [31] by chromatographic separation and confirmed by Jaggi by X-ray crystal determination [32]. In solution, each separate isomer readily undergoes equilibration to generate the original mixture. The nine isomers of the fac configuration apply to symmetrical 1:2 metal complexes having two ligands A-N-B. In the case of unsymetrical 1:2-metal complexes having the ligands A-N-B and C-N-D, six pairs of enantiomers are possible.
In the mer configuration the three coordinating atoms A, N, and C form a isosceles triangle, while in the fac system they are situated at the corners of an equilateral triangle (Figure 2.9). The distance between the atoms A and B in the equilateral triangle is smaller than that in the isosceles triangle. Figure 2.9 illustrates that azo dyes forming annelated 6/6-membered ring chelates fit the isosceles triangle better and prefer to coordinate in the sandwich manner, while dye chelates having 5/6-membered ring systems prefer to adopt the isosceles triangle geometry. A broad discussion about distances, configuration, and coloristic behavior is given in [33].
|
Figure 2.9 A) Isosceles triangle embedded in a 5/6-membered ring chelate of the Drew — Pfeiffer type. B) Isolateral triangle suiting the 6/6-memebred ring system of the sandwich type. |
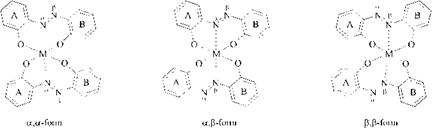
W-a, p isomerism arises from the fact that only one nitrogen atom of the azo group can act as third ligand. In the case of metallizable unsymmetrical azo compounds two isomers can be discriminated in a 1:1 metal complex [34] and three isomers in a symmetrical 1:2 metal complex [35] (Figure 2.10).
|
Figure 2.10 The three N-a, p isomers of symmetrical 1:2 metal complexes derived from unsymmetrical azo compounds, rings A and B being differently substituted. |
With mixed 1:2 metal complexes derived from two distinct unsymmetrical azo compounds four N-a, p isomers become possible. The N-a,|3 isomerism has been affirmed by NMR spectroscopy on diamagnetic 1:2 cobalt(m) complexes [35] and X-ray investigations [36].
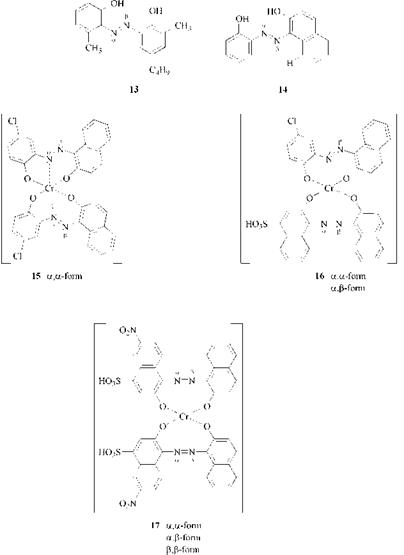
Steric factors strongly influences the relative proportions of N-aand N-13 coordination. Thus, the azo compound 13 strongly favors the N-|3 coordinated configuration as a result of the interaction of the 6-methyl group with the electron lone pair of the N-|3 atom, and the azo compound 14 coordinates predominantly through the N-a atom, because this atom interacts with the hydrogen atom in the 8-position of the naphthalene nucleus.
|
|
For example, by chromatographic techniques it can be shown that the 1:2 chromium complex 15 exhibits only one isomer (a, a form), the mixed 1:2 chromium complex 16 two (a, a and a,|3 forms), and the symmetric 1:2 chromium complex 17 three isomers (a, a, a,|3 and |3,|3 forms), assuming that these dyes exist only in the mer configuration.
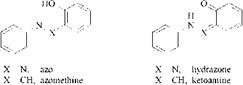
Figure 2.11 illustrates azo-hydrazone tautomerism in azo and azomethine compounds. Strongly polarized o, o’-dihydroxyazo dyes, such as 1-(2-hydroxyphe — nylazo)-2-naphthols or 4-(2-hydroxyphenylazo)-5-pyrazolones, exist predominantly in the hydrazone form. Metallizable hydrozone compounds coordinate only through the imino group due to its stronger donor property. They do not exhibit N-a,|3 isomerism, because only one hydrazone form is predominant. In contrast to the planar azo form, the donor nitrogen atom of the hydrazone group is sp3 hybridized and therefore tetrahedral. In this situation the ligand can adopt two nonplanar configurations, leading to three enantiomeric conformers in 1:2 metal complexes if both ligands exist in the hydrazone form. If one ligand coordinates in the azo and the second in the hydrazone form, two dl conformers are possible. Only one pair of enantiomers arises if both ligands are in the azo form. Thus, six isomers and their mirror images can be considered if azo-hydrazo tautomerism is present [37]. As mentioned previously in the case of N-a,|3 isomerism, this type of isomerism is also based on the assumption that the o, o’-dihydroxyazo and — azomethine compounds form only mer complexes. The fact that these conformers coexist in equilibrium means that the colors tend to rather dull shades, whereas planar metal-complex dyes, such as copper phthalocyanines and copper formazans are brighter in hue.
Chromium, cobalt and copper chelates are applied mainly in textile dyeing. The use of the less stable iron chelates is restricted to leather dyeing, and the nickel chelates are employed mostly as pigments.
|
Figure 2.11 Tautomerism in azo and azomethine compounds. |
 5 сентября, 2015
5 сентября, 2015  Pokraskin
Pokraskin 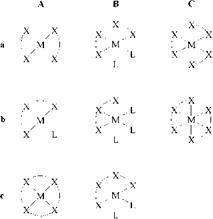

 Опубликовано в рубрике
Опубликовано в рубрике 