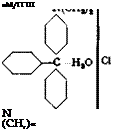
Auramine OO[60]
In an apparatus such as that shown in Figure 36a and b, a mixture of 127 grams (0.5 mole) of 4,4′-tetramethyldiaminodiphenylmethane (page 137), 32 grams of sulfur, 70 grams of ammonium chloride, and 1000 grams of pure sodium chloride, is heated to 110°C. It is essential that all of the materials be very finely ground and contain no water. During the course of 2 hours, the temperature is raised to 130° (oil bath temperature, about 25° higher) while a rapid stream of dry ammonia is introduced into the apparatus from a cylinder. Remaining traces of moisture are carried out by the ammonia, and at about 140° a vigorous evolution of hydrogen sulfide begins and continues for 10 to 15 hours, depending on the velocity of the ammonia stream. The temperature is raised to 175° over a period of 5 hours while stirring is continued, and the hydrogen sulfide is absorbed in concentrated sodium hydroxide. It is advantageous to build up a small pressure (about 0.2 atmosphere) in the apparatus (manometer!). The velocity of the ammonia stream should correspond to about 5 bubbles per second. The ammonia must be dried by passing it through a wash bottle containing 50 per cent potassium hydroxide solution and then through two towers containing sodium hydroxide sticks. (Ammonia and all amines combine with calcium chloride and hence cannot be dried with this reagent.)
When the evolution of hydrogen sulfide ceases, the reaction vessel is opened and the brownish yellow, powdery material is transferred to a large porcelain dish and mixed with 3 liters water to dissolve out the salt. The dye is then filtered off and dissolved in about 1500 cc. water at 60°C. Higher temperatures should not be employed because aura — mine is easily decomposed. The solution is filtered to remove a small residue of sulfur and Michler ketone, and is then mixed with 1 liter of the salt solution obtained as the preceding filtrate. The auramine precipitates as lustrous golden plates. The yield of pure dye is about 155 grams. It gives pure yellow shades on cotton mordanted with tannin and tartar emetic.
Technical Observations. Auramine is the most important basic yellow and is highly valued because of the extraordinarily pure tints it produces. Manufacture of the dye is done in oil heated vessels, and the heating must be very carefully
controlled since the smallest variation reduces the yield. The purity of the salt used is of great importance. Traces of calcium or magnesium chloride, which are usually present in common salt, have a deleterious effect. The best salt is the Galician rock salt which is practically chemically pure. The ammonia, dried in towers charged with sodium hydroxide, is introduced in sufficient amount to produce a pressure of 0.5 atmosphere and is circulated by pump over the stirred salt mixture. The hydrogen sulfide is absorbed and recovered as sodium sulfide to be used in reductions. In well operated plants, the yield of auramine is as much as 132 per cent, that is, 132 kilograms of pure auramine from 100 kilograms of tetramethyl — diaminodiphenylmethane. The determination of the yield is rather difficult, because only a few people are able to estimate exactly the dye strength on tanned cotton. Consequently, the dye is assayed, not by a dyeing test, but by hydrolyzing it with dilute hydrochloric acid, treating with sodium hydroxide, distilling off the ammonia into normal hydrochloric acid, and back-titrating. In many plants, however, the auramine is mixed with a known quantity of a blue basic dye, and a dyeing test is made. The resulting green tint is much easier to estimate than the pure yellow.
In addition to auramine OO, auramine G is now also manufactured, starting with monomethyl-o-toluidine. Auramine G gives purer and more greenish tints than the OO product. The product from diethylaniline is not manufactured because it precipitates, on salting out, in such a tarry condition that it cannot be worked up.
Auramine is used widely in dyeing cotton, but still more for paper. The Swedish match factories alone use eight carloads per year for coloring match boxes.
 |
Malachite Green94
 4 декабря, 2015
4 декабря, 2015  Pokraskin
Pokraskin  Опубликовано в рубрике
Опубликовано в рубрике 