The first law of adhesion states that all atoms adhere. Therefore we would expect that blocks of material placed in good contact should stick together. This idea had been studied by Tyndall in 1875 and investigated further by Budgett in 1911.3 The objective was to show that adhesion between flat plates could be larger than that expected from atmospheric pressure.
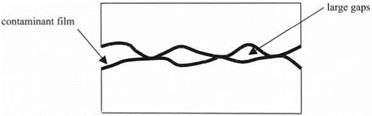
Steel blocks used as gauges in the metal working industries were highly polished to produce flatness much less than the wavelength of light, i. e. less than 100 nm. Tyndall placed these in contact by “wringing.” In other words he pressed and twisted them together in a sliding motion (see Fig. 4.1). The adhesion between the plates was then considerable and not affected much by placing the plates in a vacuum, demonstrating that atmospheric pressure was not the cause of the adhesion. However, it was clear to Budgett, when he repeated Tyndall’s observations, that the surface condition of smoothness and contamination was also vital (see Fig. 4.2). He showed that, when the surfaces were contaminated with dust, then the adhesion was reduced. More importantly, Budgett demonstrated that, when the surfaces were cleaned with solvent to remove grease and water, the blocks fell apart easily. Thus, the adhesion force was due mainly to the pulling apart of the contaminant film. McBain and Lee showed later that thin films of many materials could give a large adhesion, with strengths up to 40 MPa, when used as glue between smooth metal plates.4
|
Figure 4.1. Experiment to test adhesion of steel blocks: polish blocks, wiring together, apply force to pull apart. |
|
Figure 4.2. Model of polished metal surfaces with roughness and contaminant film |
It was clear that measuring adhesion of “flat, clean” surfaces was doomed to failure.
Budgett showed especially that the steel plates were reactive and that the contaminant moisture could generate rust-like reaction products which would also stick the plates together with large adhesion. This effect will be described in Chapter 11. Hardy and his colleagues also showed that long chain acids could give stronger adhesion, especially if time was allowed for ordering to occur.5 But the main advance came by changing the material from steel or glass to mica.
 14 сентября, 2015
14 сентября, 2015  Pokraskin
Pokraskin 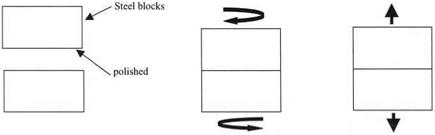
 Опубликовано в рубрике
Опубликовано в рубрике 