Heat or temperature has a considerable effect on curing, working life, and stability of adhesives. Several structural transitions can occur in adhesives during heating. Thermal analysis techniques can provide a detail road map of curing, properties, and stability of cured adhesives that has predictive utility. They also provide the ability to assess degree of cure and composition.
Thermal analysis refers to any technique for the study of materials which involves thermal control [49-52]. The techniques include the measurement of temperatures at which changes may occur, the measurement of the energy absorbed (endothermic transition) or evolved (exothermic transition) during a phase transition or a chemical reaction, and the assessment of physical changes resulting from changes in temperature. Measurements are usually made with increasing temperature, but isothermal measurements or measurements made with decreasing temperatures are also possible. Hence thermal analysis has provided important contributions in the characterization of adhesives, and a great deal has been
|
Figure 17 The 13C NMR spectrum of the reaction mixture of urea-formaldehyde concentrate with phenol under acidic conditions. |
written on this subject. It yields a ‘‘fingerprint’’ that may uniquely characterize the adhesive and assess its thermal stability. Thermal analysis data may also permit the evaluation of the kinetic parameters for the chemical changes that may have taken place during the heating process. For insoluble network adhesives, thermal techniques have been used to establish the degree and rate of cure, to study the chemical kinetics of curing reactions and the curing behavior itself, and to study degradation reactions.
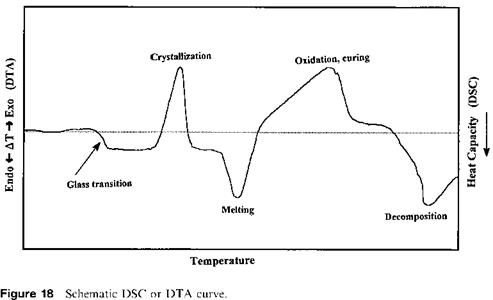
Differential thermal analysis (DTA) and differential scanning calorimetry (DSC) are techniques that monitor either the heat evolution or absorption for any reactions that are occurring in a sample. Thus, DTA/DSC thermograms reflect changes in the energy of the system under investigation—changes that may be chemical or physical in origin. The technique is particularly useful for adhesives because polymerization or structural changes are almost invariably accompanied by energetic effects so that curing, crystallization, melting and other reactions all show characteristic DTA/DSC thermograms (see Fig. 18). The use of dynamic mechanical thermal analysis (DMTA), however, is not involved in heat change; it is a technique that measures molecular motion in adhesives to obtain a temp — erature/modulus relationship. These thermal analysis techniques are discussed below. 1
|
|
|
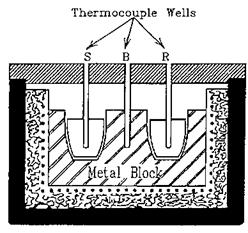
Figure 19 The construction of a DTA apparatus. |
illustrates the principle. Two small crucibles, placed in suitably shaped cavities in a metal block, contain the sample (S) and the reference (R). The two junctions of a thermocouple are inserted into the crucibles so as to give directly the temperature difference between them. A separate thermocouple is placed in cavity B to measure the temperature of the block. The entire assembly is then heated under the control of a linear programmer. With constant heating, any transition or thermally induced reaction in the sample will be recorded as a peak or dip in an otherwise straight line. An endothermic process will cause the thermocouple junction in the sample to lag behind the junction in the reference material, and hence develop a voltage, whereas an exothermic event will produce a voltage of an opposite sign. It is customary to plot exotherms upward and endotherms downward.
|
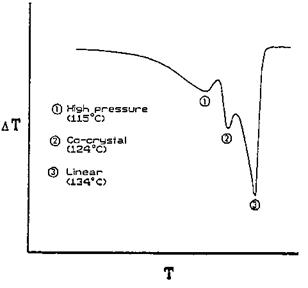
Figure 20 Typical DTA thermogram of a linear high-pressure polyethylene blend. |
Conventional DTA can give good qualitative data about temperatures and signs of transitions, but it is difficult to obtain quantitative information about the sample or the heat of transition. Figure 20 shows a typical DTA thermogram of a linear high-pressure polyethylene blend [53]. This polymer, upon heating, undergoes three phase changes from its high-pressure form (115°C) to co-crystalline form (124°C) to a linear form (134°C). The 115°C peak was associated with the high-pressure polyethylene, whereas the 134°C peak was shown to be proportional to the linear content of the system. Recently, it was shown that there is reasonable correlation between the gelation time and the temperature corresponding to the peak of the isotherm on curves for an epoxy resin system [54]. The DTA method has yielded accurate kinetic results for a PF resin system, in addition to providing an insight to the actual chemistry of the curing process [55]. Orrell and Burns [56] used DTA to investigate phenol HMTA and novolak HMTA reactions.
 10 июля, 2015
10 июля, 2015  Malyar
Malyar 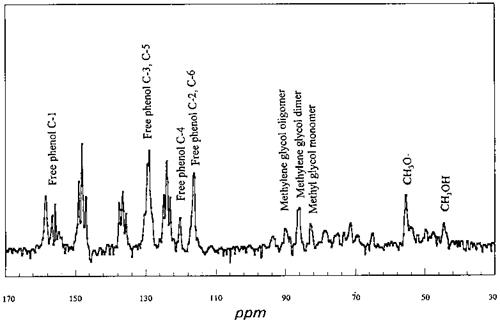
 Опубликовано в рубрике
Опубликовано в рубрике 